The disease and the virus
Viral Encephalopathy and Retinopathy is a viral disease affecting more than 50 fish species, both wild and farmed (sea bass, grouper, sea bream, striped jack, flat fish etc.)(OIE, 2016). The disease causes neurological symptoms and increased mortalities. The disease occurrence has a seasonal pattern, with most outbreaks during the summertime, because of the higher temperatures of the water, which favors virus replication. Fish fry and larvae seem to be most susceptible, and in this age-group the disease can cause up to 100% mortality. Transmission of the disease can occur both horizontal (from fish to fish or equipment/feed to fish) and vertically associated (from brood stock to progeny), and the virus is very persistent in the environment, making spread with vectors (such as boats, feed and equipment) possible. As the name suggest, the disease manifest in the central nervous system causing histopathological necrosis in the brain and retina. Affected fish will either show clinical symptoms such as loss of buoyance control, spiral swimming pattern and darkening of the skin, or die before symptoms appear. Historically, the disease has also been referred to as Viral Nervous Necrosis (VNN) or Fish Viral Encephalitis (FVE), amongst other names; however, the official name, as recognized by the OIE (Office International des Epizooties) is Viral Encephalopathy and Retinopathy (VER)(OIE, 2016). VER is caused by a small non-enveloped positive sense single stranded RNA virus called betanodavirus. The virus particle is 25-35 nm in diameter and has a capsid (a viral capsule) with protruding spikes, making the virus able to enter the fish cells.
Biosecurity
Because of the high persistency in the environment, the ability to transmit horizontally, and the possibility of infecting wild fish, serving as a reservoir for virus, the virus is difficult to prevent with biosecurity measures alone. That being said, high biosecurity measures should always be applied where possible to prevent VER and other diseases. These include, but are not limited to (Costa & Thompson, 2016; OIE, 2016);
Facilities:
- Regular cleaning and disinfection of boats, equipment, tanks, filters.
- Protocols for changing of clothes and equipment when moving around between different units on the farm, to avoid internal spread. Especially when handling fry and larvae, which are most susceptible.
- Proper cleaning of tanks, hatching facilities etc. between each hatch and when moving fish around.
- Avoid using raw fish in feed
- Reduce stress, especially around spawning i.e. by providing adequate food and decreasing stocking density
- If an outbreak has been diagnosed, thorough cleaning and disinfection, often several times, is essential to avoid re-infection when introducing new fish.
Animals:
- When importing fish, eggs or larvae to the farm, always ask the sender for documentation of disease-free status, and if possible, keep the received fish in quarantine upon arrival, to avoid introducing new diseases.
- Disinfect fertilized eggs and/or rearing water.
- Regular health screening of the stock, to catch the infection early and prevent vertical transmission.
- Screening of ovarian biopsies, eggs and sperm from brood stock.
- Screening of blood from spawners for detection of antibodies against VER
- Screening of hatched larvae
Vaccination
A vaccine works by training the immune system to fight a given pathogen (i.e. bacteria or virus), so that when the animal encounters the pathogen, the immunological defense mechanisms are ready to fight and combat the pathogen. A successful vaccine will therefore prevent outbreak and spread of the disease, which improves both animal welfare and the economy of the farmer. Disease prevention by vaccination should always be supported by correct biosecurity measures to keep the exposure to the pathogen as low as possible.
There are marketed vaccines for sea bass protecting against some bacterial diseases (i.e. Vibrio), however until recently there has been no available vaccine to protect against VER. When developing vaccines for fish there are some factors that is important to consider
- Price – the price must reflect the fact that several thousand fish are to be vaccinated, and if it is too expensive, the farmer might choose not to vaccinate. This demands for a cheap and high yield production to keep the price down.
- Delivery route and dosage – practical things as how the vaccine is administered (oral, bath or injection?) and when the fish can receive the vaccine is important too. VER is a disease that causes mortality in the early life stages of the fish, and therefore the ideal vaccine should be able to protect very young fish.
There are different ways of designing vaccines. The oldest and probably widest used are the use of inactivated pathogens to stimulate the immune system. Inactivated vaccines are made by killing the pathogen i.e. by formalin or heat-treatment and then using the killed pathogen in the vaccine. This type of vaccine is usually very safe, however it can be difficult to get a good protective immune response against the killed pathogen. Therefore, helping-ingredients, also called adjuvants, are added, such as oil or alum. Very recently, an inactivated vaccine against VER for sea bass has been licensed by the company Pharmaq for use in selected Mediterranean countries. It is to be given by intraperitoneal injection from when fish are at a size of 12 g.
A variation of the inactivated vaccine is the attenuated vaccine – in this case, the pathogen is not completely killed, however weakened enough to not cause disease. This usually gives a better immune response than the killed vaccine, however there is a risk of the pathogen re-gaining the ability to cause disease (“reversion to virulence”), which can have fatal consequences.
Advancements in vaccine production has allowed for new vaccine-designs without the use of the whole pathogen, therefore limiting the risk of reversion to virulence (2nd generation vaccines). These designs can include sub-units (small parts) of the pathogen or toxins from the pathogen (in case of bacteria). These types of vaccines has been experimentally developed against betanodavirus and tested, however with varying results (Coeurdacier, Laporte, & Pepin, 2003).
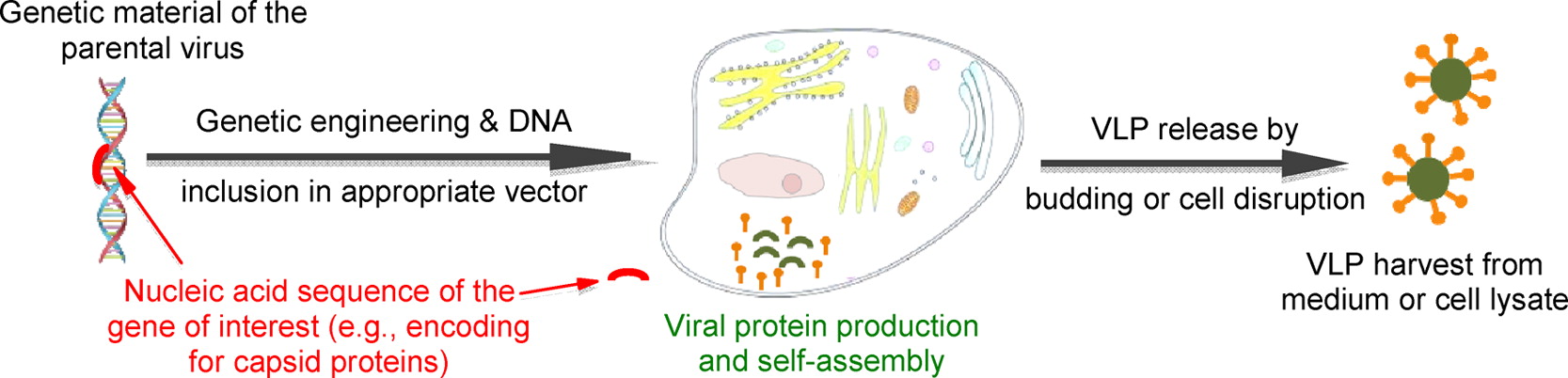
Even more advanced are the 3rd generation vaccines, such as DNA vaccines and recombinant protein vaccines. These types of vaccines has also been developed and tested against betanodavirus. The vaccine that we work with in WP 4 of the MedAid is a 3rd generation “Virus-like Particle” or VLP vaccine. It is a prototype made by the German biotech company W42 (www.w42biotechnology.de) and is still only used experimentally. The vaccine is made by incorporating the gene sequence of the main viral protein (the capsid protein) a yeast cell. Upon activation, the yeast cell culture will produce the viral capsid protein, which will auto-assembly into particles that look like the virus, but without being infectious, because they lack the genetic material of the virus (figure 1). This type of vaccine has been tested against betanodavirus both in sea bass (Thiéry et al., 2006) but also in other species (i.e. grouper) (Chien, Wu, & Lin, 2018) with promising results. Working with the VLP in the MedAid framework in collaboration with W42, we will optimize the vaccination strategy in terms of delivery route, vaccine dose, adjuvant, and time (fish age) of vaccination. Once an optimal schedule has been establish under experimental conditions, we will test it in the field on farms experiencing problems with VER and hereby determine the applied potential of this 3rd generation vaccine for disease prophylaxis in Mediterranean fish farms.
![]() Complete article with references
Complete article with references
 Author:
Author:
Sofie Barsøe
Unit for Fish and Shellfish Diseases,
National Institute of Aquatic Resources,
Technical University of Denmark

